Article title: Cardiolipin dynamics promote membrane remodeling by mitochondrial OPA1
Authors: Sirikrishna Thatavarthy, Luciano A. Abriata, Fernando Teixeira Pinto Meireles, Kelly E. Zuccaro, Akhil Gargey Iragavarapu, Gabriela May Sullivan, Frank R. Moss III, Adam Frost, Matteo Dal Peraro, and Halil Aydin
Journal: Nature Communications (Link to Article)
Background
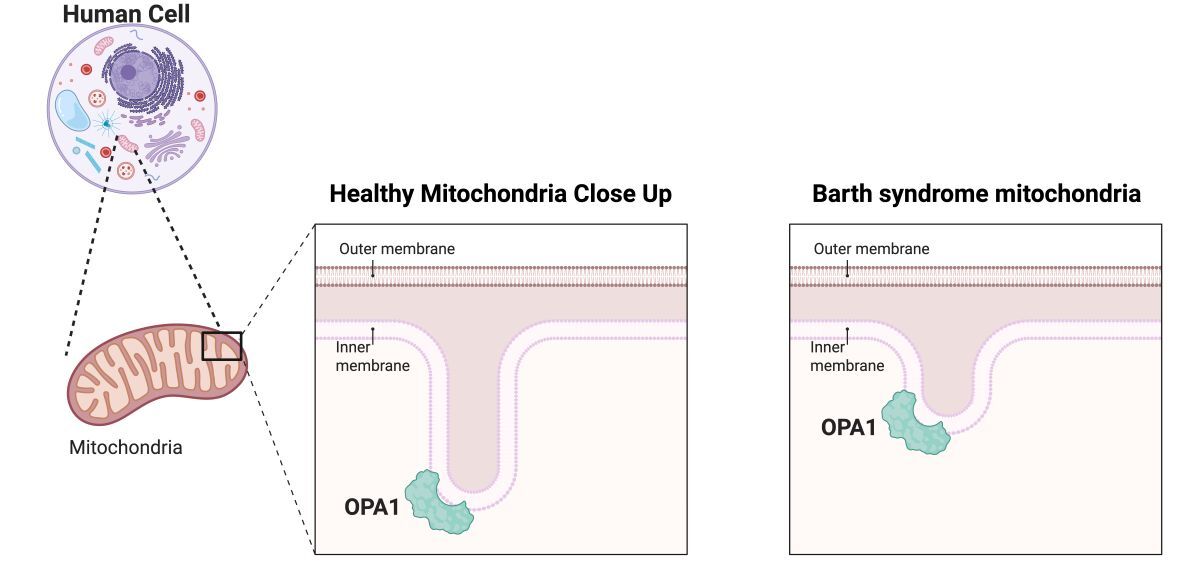
Mitochondria are specialized sub compartments in cells that are responsible for making energy—you may have learned that they are the “powerhouse of the cell”. In Barth syndrome, mitochondria cannot make enough energy for the body’s cells to do the work they need to do for energy-demanding organs like the heart and skeletal muscle. While we understand that Barth syndrome is characterized by low energy, we still do not understand all of the reasons why the energy is low. Barth syndrome is caused by mutations in a gene called TAFAZZIN. TAFAZZIN has a very important job in all mitochondria—it is responsible for converting a fat building block (monolysocardiolipin or MLCL for short) into its final form, cardiolipin. Cardiolipin is one of the primary building blocks of the internal mitochondrial scaffolding, also called a membrane. In Barth syndrome, TAFAZZIN cannot make cardiolipin and instead MLCL piles up in the membrane. This MLCL pile up changes the way mitochondria are shaped, which changes how the energy-making machinery interact with each other, ultimately making it harder to produce energy.
What
While we know mitochondria are not the right shape in people affected by Barth syndrome, we do not understand why having MLCL instead of cardiolipin in the mitochondrial membrane causes them to be misshapen.
How
Now, in a new publication in the leading scientific journal Nature Communications, a team of scientists have described important details about how cardiolipin helps keep mitochondria healthy and working properly. The research team, led by Dr. Halil Aydin (a 2024 grant recipient from Barth Syndrome Foundation), used advanced computer simulations and powerful microscopes to actually visualize exactly how cardiolipin interacts with a protein called OPA1, which is plays a vital role in giving mitochondria their shape. They found that cardiolipin molecules cluster around OPA1 like magnets and promote additional OPA1 proteins to assemble together, promoting physical changes in the mitochondrial membrane shape. When people have Barth syndrome, however, the MLCL pileup makes the membranes too stiff for OPA1 to reshape the mitochondrial membrane, which may underlie why mitochondria are misshapen in Barth syndrome patients.
Why
This research demonstrates how the Barth Syndrome Foundation’s continued investment in basic science can lead to important findings with clinical relevance. The Barth Syndrome Foundation partly funded Dr. Aydin's efforts and the resulting study directly now fills in critical gaps in our understanding of how genetic changes in TAFAZZIN cause the physical problems patients experience (heart weakness, muscle fatigue).



