Article title: Integrated multi-omics mapping of mitochondrial dysfunction and substrate preference in Barth syndrome cardiac tissue
Authors: Bauke V Schomakers, Adriana S Passadouro, Maria M Trętowicz, Pelle J Simpson, Yorrick R J Jaspers, Michel van Weeghel, Iman Man Hu, Cathelijne M E Lamboo, Denise Cloutier, Barry J Byrne, Jan Bert van Klinken, Paul M L Janssen, Sander R Piersma, Connie R Jimenez, Frédéric M Vaz, Gajja S Salomons, Jolanda van der Velden, Riekelt H Houtkooper, and Signe Mosegaard
Journal: EMBO Molecular Medicine (Link to Article)
This research was supported in part by a Barth Syndrome Foundation grant to Dr. Houtkooper in 2019.
Background
Barth syndrome is an ultra-rare genetic condition that affects mostly boys and men. It is caused by a change in a gene called TAFAZZIN and impacts the heart, skeletal muscle, immune system and gastrointestinal tract. In healthy cells, you can imagine TAFAZZIN as an important protein on a factory line that ensures the final version of a special fat called cardiolipin is made. Cardiolipin is very important in cells as it provides structural support for the energy-making machinery within each cell’s power plants, also called mitochondria.
In Barth syndrome, when the TAFAZZIN isn't made or doesn’t work properly, the body can't make mature cardiolipin. Instead, an unfinished or precursor version of cardiolipin called monolysocardiolipin or MLCL for short, is made and piles up. This traffic jam of MLCL decreases the amount of energy made by mitochondria. Because the heart is one of the hardest-working muscles in the body — constantly beating, never resting — its function is dependent on well-functioning mitochondria for energy. When mitochondria cannot produce enough energy, the heart suffers. This is why heart disease is one of the most prominent manifestations of Barth syndrome. While we understand the end result (less energy made by cells), we do not yet know all of the key players involved in this process. If we investigate who the key players are, we might uncover new targets for therapy development for the treatment of Barth syndrome.
What
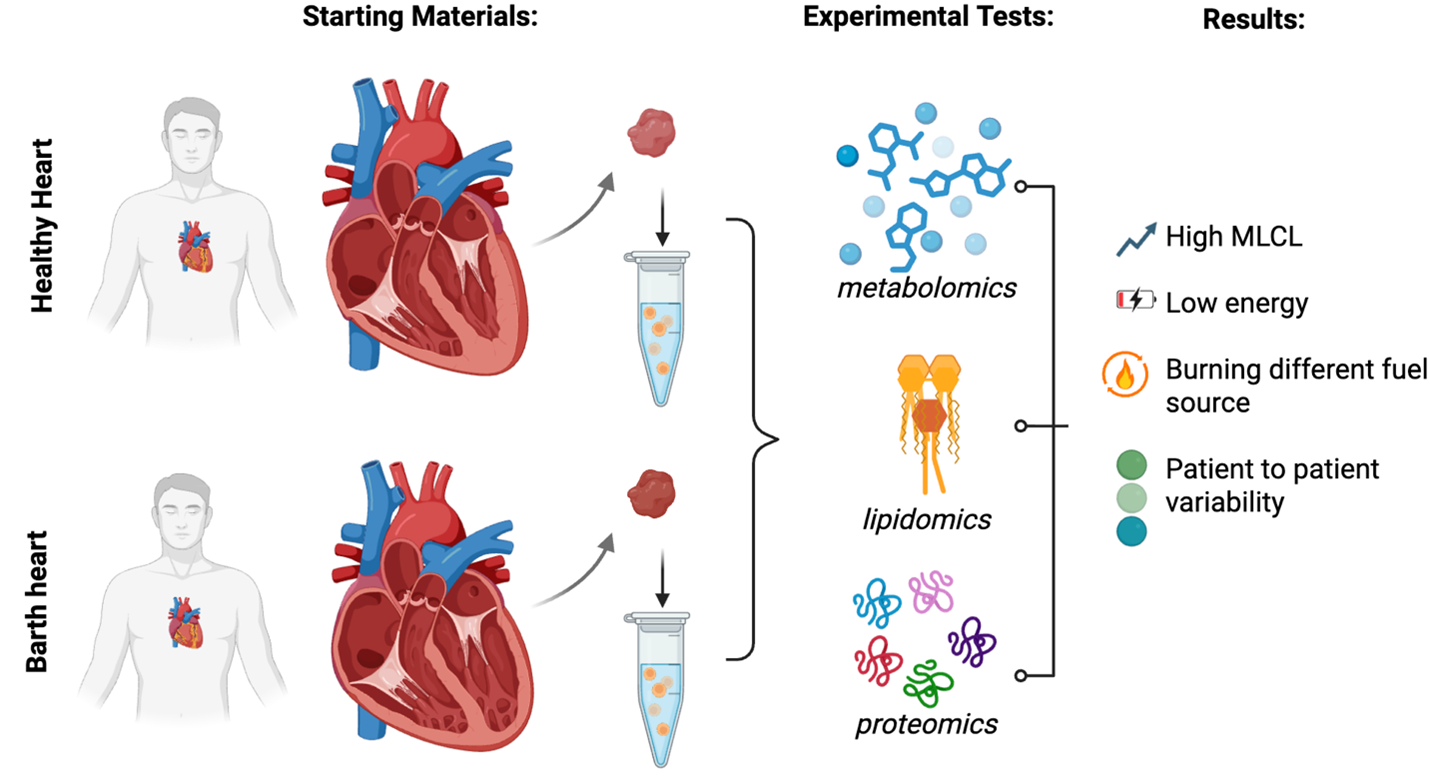
Recently, a team of researchers, including BSF Scientific and Medical Advisory Board member Dr. Riekelt Houtkooper, published a paper in EMBO Molecular Medicine that took a detailed look inside the heart using heart tissue donated by Barth affected individuals. This research was supported in part by a Barth Syndrome Foundation grant to Dr. Houtkooper in 2019. These Barth hearts were then compared them to heart tissue from 24 people without heart disease. Two of the Barth heart samples came from patients who had passed away, while three came from affected individuals who received a heart transplant.
The research team used three powerful scientific tests that were performed in the same tissues at the same time to study the hearts of Barth affected individuals: metabolomics to look at small molecules called metabolites, lipidomics to look at different fats, and proteomics to look at proteins. Using all three tools together on the same samples gave the researchers a more complete picture of how BTHS hearts differ from healthy hearts.
How
Here is a summary of the major findings:
- Lipids like cardiolipin are disrupted in Barth syndrome. Barth syndrome heart tissue had much less mature cardiolipin and a large buildup of the unfinished or precursor form (MLCL). This confirms that the experiment performed was valid and that these changes are clearly visible in heart tissue — not just in blood tests used for diagnosis.
- The heart's power plants are struggling. Mitochondria serve as a power plant for each cell. They generate energy by running a series of protein "machines" called the electron transport chain, which you can think of like a factory line. All parts of the factory line need to be there in the right order and amount to produce energy effectively. In BTHS hearts, the research team found that many of the proteins that make up the energy factory line were reduced. This means the heart is having a very hard time producing the energy it needs.
- The heart is changing how it gets fuel. A healthy heart gets most of its energy by burning fats via a process called fatty acid oxidation. In BTHS hearts, this fat-burning process appeared to be significantly impaired. To try to compensate, the heart appeared to be shifting toward burning sugars (glucose) instead — a less efficient fuel source. This kind of "fuel switch" is also seen in other forms of heart failure and is considered a sign that the heart is struggling.
- Signs of heart remodeling and stress. In the hearts collected after a patient passed away (versus those collected at the time of transplant), researchers found higher levels of warning sign signals, including a buildup of branched-chain amino acids (building blocks of protein that pile up when the heart is under stress), elevated levels of fats like ceramides and sphingosines, which can be toxic to heart cells, and changes in the structural proteins that make up the heart muscle itself
- Each patient is different. One of the important takeaways from this study is that even though all five patients have BTHS, their hearts don't all look identical at the molecular level. While the cardiolipin changes were consistent across all patients, other features varied from person to person. This may help explain why BTHS can look so different from one person to the next.
Why
Why does this research matter for our community?
This study was funded in part by Barth Syndrome Foundation and highlights how continued investment in research can yield important molecular and clinical insights. The findings in this paper will be a vital stepping stone to better understanding how Barth syndrome directly affects the heart as it gives us the most detailed molecular map of a BTHS heart to date. Previous studies mainly looked at blood, skin cells, or animal models while this study examined human hearts from Barth affected individuals.
It validates what we already suspected and reveals new clues. The study confirmed known hallmarks of BTHS (like cardiolipin changes and mitochondrial dysfunction) are present in human hearts but also gave rise to new details about how the heart tries to cope — and where those coping strategies fall short.
It could point toward new treatment targets. By mapping out which proteins, fats, and metabolites are most disrupted, researchers now have a clearer list of potential targets for future therapies.