Program Overview
Applications for the 2026 cycle are now closed.
All applications must be submitted via BSF's Survey Monkey Apply portal.
The Barth Syndrome Foundation (BSF) and our International Affiliates welcome innovative applications that address the basic, translational, and clinical research challenges of Barth syndrome. BSF seeks to provide seed grant funding to young and established investigators to help catalyze generation of preliminary data that are required for successful follow-on funding from institutions like the National Institutes of Health.
Applicants must be independent investigators (e.g. faculty appointment). Pre- and postdoctoral applicants are encouraged to apply to the American Heart Association & BSF Predoctoral and Postdoctoral Fellowship program (Deadlines: 9/3/25 and 9/4/25, respectively). For questions, please email our AHA colleagues via apply@heart.org
Key Dates:
- Full Applications - Due Friday, October 31, 2025
- Grantees Announced and Earliest Award Start Date - March 2026
Application Specifics:
- up to $100,000 USD maximum Total Cost, 1-3 years in duration.
- Researchers seeking Scientific and Medical Advisory Board (SMAB) feedback are encouraged to email the BSF staff for support
Application and Review Process
BSF administers a competitive grant process, with a peer review stage that involves two (2) members of the BSF Scientific and Medical Advisory Board (SMAB) and one (1) external reviewer with assay or topical experience relevant to the submitted application. More details of our program can be found in our Application Guidelines and Requirements document.
Full Application - Components of the Full Application include detailed information about the specific aims, significance, research strategy and methods, budget, as well as the personnel and facilities that would support a proposal.
Evaluation Criteria - All submitted applications will be scored on the basis of three (3) criteria:
- Importance and Impact - the extent to which the research outlined could make a significant contribution to the understanding of Barth syndrome or the treatment of the disorder.
- Feasibility - the qualifications of the investigator(s) to carry out the proposed work based on expertise, experience, and commitment; as well as the availability of all the elements required for the project (such as technology, reagents, and research participant engagement and recruitment).
- Prospect for Long-term Funding - the likelihood of subsequent NIH or other grant funding institution to support follow-on work.
Decision to Fund - Based on the final recommendations of the SMAB, the BSF Board of Directors will make the final decision of whether or not to fund submitted applications.
Historical Funding
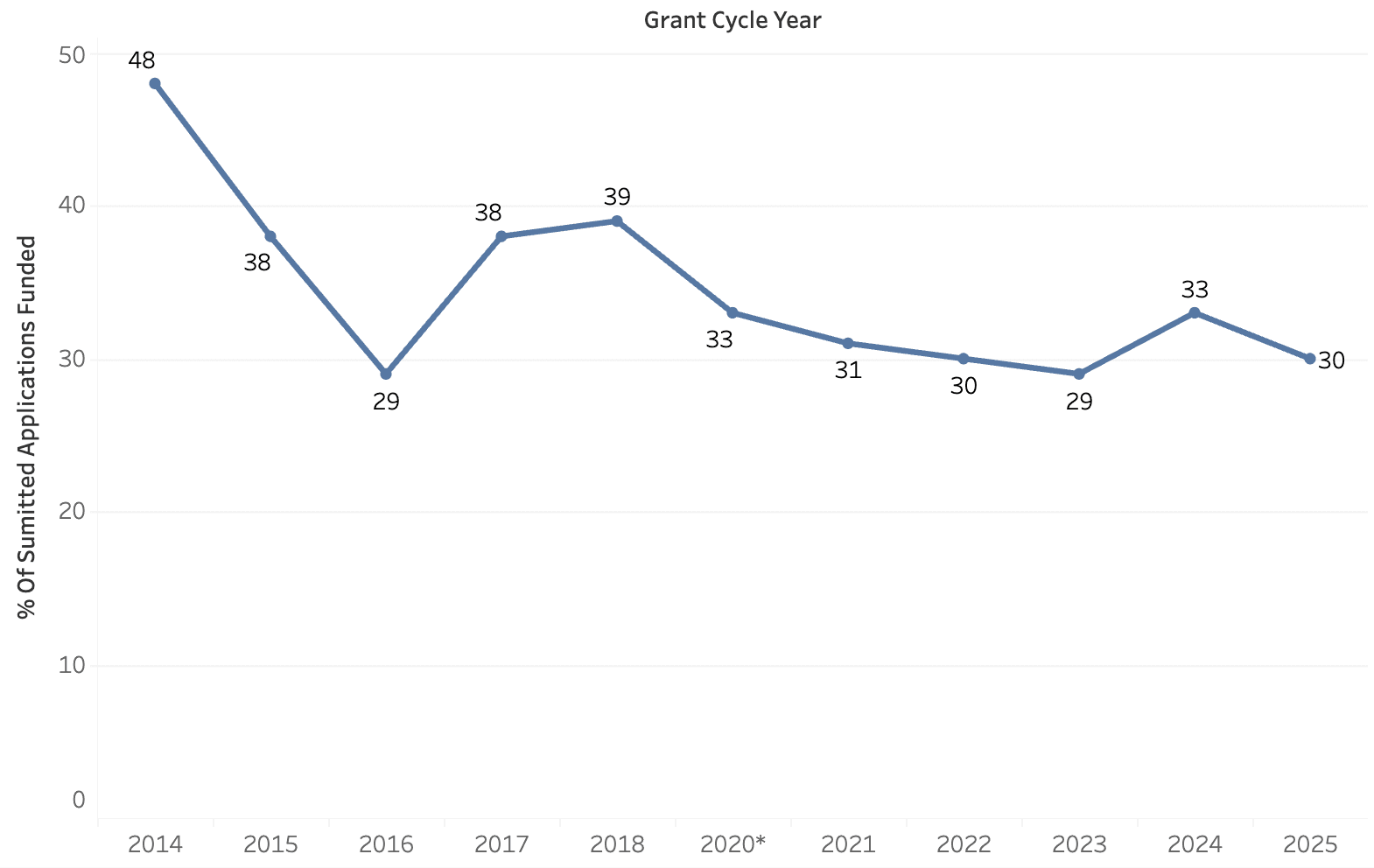
* BSF modified its grant cycle dating convention to follow the year upon which research funds are disbursed. Previously, the Foundation referred to the year in which the application was submitted.
BSF Research Staff Contact Information
For science-related inquiries, please contact:
Lindsay Marjoram, PhD
Director of Research
Barth Syndrome Foundation, Inc.
lindsay.marjoram@barthsyndrome.org
For application process questions, please contact:
Melissa Huang, PhD
Research Associate, Research Engagement & Assets
Barth Syndrome Foundation, Inc.
melissa.huang@barthsyndrome.org
Grant Program Application and Guidelines
Last updated 8/26/2024




